A project in which Cranfield University played a vital role has redefined the scientific unit of temperature, the kelvin. The kelvin is one of 7 base units in the International System of Units (SI), the world’s most widely used system of measurement.
Researchers at the National Physical Laboratory (NPL) in Teddington, have, for the past six years, been undertaking an experiment as part of a radical revision of the whole SI system that has now yielded the most accurate temperature measurement ever made and pushed science and engineering to the limit.
Researchers have been painstakingly working to try and define the kelvin via an unchangeable fixed constant of nature and, in doing so, make it a more reliable measurement. Their solution is to use the ‘Boltzmann constant’ named after Ludwig Boltzmann, as the base for defining temperature. This is one of the fundamental constants of nature and relates energy at the individual particle level with temperature.
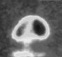
An accurate measurement for the Boltzmann constant had to be calculated. Doing so required a special acoustic resonator – a hollow ellipse shaped copper chamber made and measured to a phenomenal level of accuracy and with a finish of a few nanometres roughness, so mirror smooth.
NPL enlisted the help of Cranfield’s world renowned precision engineering expertise and Paul Morantz and colleagues created and measured the copper resonator used in the experiment more accurately than anyone else has been able to do.
The importance of the whole project was the accuracy of the surface, which meant the resonator had to be measured in six independent ways in order to verify its shape and size to produce a measurement of its dimensions with an uncertainty of just a few nanometres – smaller than many molecules.
During the experiment, the resonator was cooled to the triple point, injected with argon gas and the speed of sound was measured to calculate the average speed of the argon molecules and the average amount of kinetic energy present. From this the Boltzmann constant was calculated with extremely high accuracy and to the lowest ever uncertainty.
Cranfield University has played a key role in helping to define all international scales of temperature; this is one of the proudest achievements of Cranfield’s Precision Engineering Institute.